 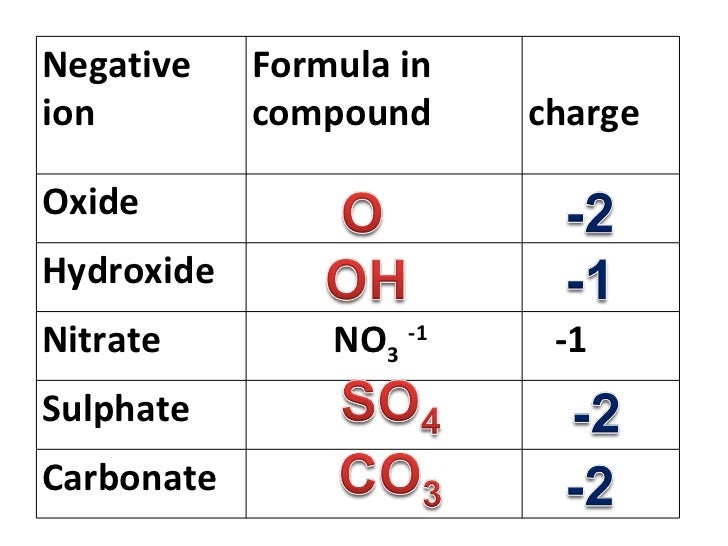 
It is a catalyst for the synthesis of sodium amide from a solution of sodium in ammonia: 2 NH 3 + 2 Na → 2 NaNH 2 + H 2Ĭertain clays impregnated with ferric nitrate have been shown to be useful oxidants in organic synthesis. The compound can be prepared by treating iron metal powder with nitric acid, as summarized by the following idealized equation: Fe + 4 HNO 3 + 7 H 2O → Fe(NO 3) 3(H 2O) 9 + NO Applications įerric nitrate has no large scale applications. Another method for producing iron oxides from this nitrate salt involves neutralizing its aqueous solutions. When this solution is heated to near boiling, nitric acid evaporates and a solid precipitate of iron(III) oxide Feģ appears. When dissolved, iron(III) nitrate forms yellow solutions. It is for example, a standard precursor to potassium ferrate K 2FeO 4. Iron(III) nitrate is a useful precursor to other iron compounds because the nitrate is easily removed or decomposed. hexahydrate ( x=6), more precisely hexaaquairon(III) trinitrate, (NO 3) 3, where the Fe 3+ ion is coordinated to six water molecules in octahedral configuration. 
pentahydrate ( x=5), more precisely penta-aqua nitratoiron(III) dinitrate, (NO 3) 2, in which the Fe 3+ ion is coordinated to five water molecules and a unidentate nitrate anion ligand in octahedral configuration.H 2O, has complex cations wherein Fe 3+ is coordinated with two nitrate anions as bidentate ligands and three of the four water molecules, in a pentagonal bipyramid configuration with two water molecules at the poles.tetrahydrate ( x=4), more precisely triaqua dinitratoiron(III) nitrate monohydrate, NO 3.This compound is the trinitrate salt of the aquo complex 3+. 9H 2O, which forms colourless to pale violet crystals.Iron(III) nitrate is deliquescent, and it is commonly found as the nonahydrate Fe(NO 3) 3 The hydrates are all pale colored, water-soluble paramagnetic salts. Most common is the nonahydrate Fe(NO 3) 3. So long as they are used for that purpose only, and not taken to mean that atoms in covalent species actually have the large charges oxidation numbers often imply, their use is quite valid.Iron(III) nitrate, or ferric nitrate, is the name used for a series of inorganic compounds with the formula Fe(NO 3) 3. Even though this may (and indeed should) make you suspicious of the validity of oxidation numbers, they are undoubtedly a useful tool for spotting electron-transfer processes. Instead, there are covalent bonds and electron-pair sharing between nitrogen and oxygen in both species, and nitrogen has certainly not lost its valence electrons entirely to oxygen. The nitrogen atom in NO 3 – does not really have a +5 charge which can be reduced to +4 in NO 2. Copper was oxidized because its electrons were accepted by an oxidizing agent, nitrogen (or nitrate ion).Īlthough they are useful and necessary for recognizing redox reactions, oxidation numbers are a highly artificial device. The nitrogen was reduced by electrons donated by copper, and so copper was the reducing agent. Each nitrogen gained one electron, so 2 e – were needed for the 2 NO 3 –. The oxidation number of nitrogen went down from 5 to 4, and so the nitrogen (or nitrate ion) was reduced. Since the oxidation number of copper increased from 0 to +2, we say that copper was oxidized and lost two negatively charged electrons. Applying the oxidation number rules to the following equation, we have Oxidation corresponds to increasing the oxidation number of some atom. As a general rule, reduction corresponds to a lowering of the oxidation number of some atom. This arbitrarily assigned gain of one electron corresponds to reduction of the nitrogen atom on going from NO 3 – to NO 2. In NO 2, on the other hand, the nitrogen has an oxidation number of + 4 and may be thought of as having one valence electron for itself, that is, one more electron than it had in NO 3 –. This arbitrary assignment corresponds to the nitrogen’s having lost its original five valence electrons to the electronegative oxygens. This is done by assigning oxidation numbers to each atom before and after the reaction.įor example, in NO 3 – the nitrogen is assigned an oxidation number of +5 and each oxygen an oxidation number of –2. In order to be able to recognize redox reactions, we need a method for keeping a careful account of all the electrons. Redox reactions may involve proton transfers and other bond-breaking and bond-making processes, as well as electron transfers, and therefore the equations involved are much more difficult to deal with than those describing acid-base reactions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
